Short term safety and pharmacokinetic profile of asenapine in older patients with psychosis
Статьи Опубликовано в журнале:«INT J GERIATR PSYCHIATRY»; 2011; стр. 1-11.
Steven L. Dubovsky1, Colleen Frobose2, Phillip Phiri2, Rik de Greef3 and John Panagides4
2Merck, Rahway, NJ, USA
3Merck Sharp & Dohme, Oss, Noord-Brabant, The Netherlands
4Merck & Co, Inc, Summit, NJ, USA
Objectives: The aim of this study was to assess the short-term tolerability of two titration schedules of sublingual asenapine in older patients with psychosis, not associated with organic brain disease, and to compare asenapine pharmacokinetics in older patients versus younger adults with schizophrenia.
Methods: Patients ?65 years with psychosis without dementia were randomized for 6 weeks to two dose-escalation regimens: 2 days at 2 mg twice daily (BID), 2 days at 5 mg BID, and 10 mg BID thereafter (slow escalation); or 4 days at 5 mg BID and 10 mg BID thereafter (rapid escalation). Clinical and pharmacokinetic assessments were performed in each group.
Results: Of 122 randomized patients, 76 (62.3%) completed the trial. The incidence of treatment-emergent adverse events (AEs) was comparable (72.1%) with both regimens. The most frequently reported AEs were hypertension, headache, and somnolence; incidence of extrapyramidal symptom-related AEs was 5.7%. Mean end point weight change was 0.4 kg. For asenapine 5 and 10 mg BID, median times to maximum concentration were 1.00 and 1.06 h, respectively; maximum concentrations (Cmax) were 4.73 and 7.93 ng/mL; areas under the concentration versus time curve (0-12 h; AUC0-12) were 32.1 and 56.3ng-h/mL.
Conclusions: Despite 12-30% increases in asenapine Cmax and AUC0-12 in older patients compared with previously published findings in younger schizophrenia patients, possibly as a result of slower drug clearance, asenapine was generally well tolerated during both dose-escalation schedules. No dose adjustment appears to be necessary in older patients. Copyright © 2011 John Wiley & Sons, Ltd.
Key words: asenapine; older; psychosis; safety; schizophrenia; pharmacokinetics
Introduction
By 2030, the population of adults ?65 years in the USA is expected to increase to about 69.4 million, with estimates that about 15 million ofthese individuals will have psychiatric illnesses, including psychotic disorders such as schizophrenia (Jeste et al., 1999). Among adults ?65 years, the 1-month prevalence of schizophrenia has been estimated at 0.1% in community samples (Regier etal., 1988). However, the true prevalence maybe higher because some studies in older adults do not include late-onset schizophrenia (Hybels and Blazer, 2003).
The increased number of older adults with psychotic disorders that is likely to occur as the population ages creates a need for well-tolerated antipsychotic medications for older patients. In a cross-sectional population-based study of 1.4 million Canadian residents, the prevalence of antipsychotic use among adults ?65 years increased from 2.2 to 3.3% between 1993 and 2002, with atypical antipsychotics accounting for 82.5% of all antipsychotics dispensed in 2002 (Rapoport et al., 2005). Although newer agents have generally been used preferentially, older individuals are still at increased risk for extrapyramidal side effects (EPS), stroke, and sudden cardiac death (Douglas and Smeeth, 2008; Uchida et al., 2009), although the data are mixed (Herrmann et al., 2004; Gill et al., 2005). In general, changes in drug distribution, metabolism, and excretion can increase drug exposure in older patients (Klotz, 2009); age-related receptor changes, polypharmacy, and comorbidities can also increase all adverse effects in this population (Gareri et al., 2006; Klotz, 2009).
Asenapine is an atypical antipsychotic drug indicated for treatment of schizophrenia and for acute treatment of mania (2010b) (2010a). Asenapine is extensively metabolized, primarily through glucuronidation by uridine 5'-diphospho-glucuronosyl-transferase and oxidative metabolism by cytochrome P450 (CYP) 1A and 3A isoenzymes (2010b). Two of the major metabolites are N-glucuronide and N-desmethylasenapine (2010b). Asenapine appears to be well tolerated in both short-term (6 weeks) and long-term (26-52 weeks) schizophrenia trials (Potkin et al., 2007; Kane et al., 2010; Schoemaker et al., 2010), with a low propensity to induce extensive weight gain and minimal impact on total cholesterol and fasting glucose in younger patients (Potkin et al., 2007; Kane et al., 2010). Further, EPS with asenapine appear to be less common than with haloperidol and risperidone (Potkin et al., 2007; Kane et al., 2010); increased prolactin is less common than with risperidone (Potkin et al., 2007). However, in these studies, there were few older patients, so an assessment of age-related changes in safety and tolerability age could not be performed. Because the standard approach to medication escalation in older patients is to "start low and go slow," it is appropriate to study the relationship of adverse effects to dosage protocols in the older patients.
Although specific relationships between pharmacokinetics (PK) and drug efficacy and safety have not yet been identified for asenapine, PK alterations are known to alter effects of other drugs and could be relevant for asenapine. For instance, slower metabolism could result in drug accumulation and a subsequent increase in adverse effects. Conversely, increased metabolism could increase drug clearance and result in reduced efficacy.
In a pooled population PK analysis of asenapine in mixed age patients with schizophrenia or bipolar disorder, older age was associated with decreased asenapine clearance, resulting in exposure in 60-year-old patients being 26% higher than in 20-year-old patients (Prohn et al., 2009). Because age-related PK alterations might influence the clinical profile of asenapine, it is important to examine asenapine PK further in older patients. The primary objective of the present study was to evaluate the safety and tolerability profile of asenapine between two titration schedules in patients with psychotic symptoms who were ?65 years old. An additional objective was to assess the PK profile of asenapine in these patients. In order to suggest possible clinical implications and to generate hypotheses for further studies, the observed PK profile was compared with previously published findings from a younger population of individuals treated with asenapine (Chapel et al., 2009).
Methods
Study design
This randomized, parallel group, multiple-dose, 6-week study (A7501021; NCT00281320) was conducted in the USA, the Czech Republic, Russia, the Ukraine, and the Philippines between January 2006 and December 2008. The study was conducted in accordance with principles of Good Clinical Practice and was approved by the appropriate institutional review boards and regulatory agencies. All patients gave written informed consent.
Study population
Patients were men or women ?65 years with scores of ?4 on ?1 of five prespecified positive and negative syndrome scale (PANSS) items (delusions, hallucinatory behavior, excitement, hostility, and poor impulse control), a PANSS total score >50, and a Clinical Global Impression-Severity of Illness (CGI-S) score of ?3 at screening. Key exclusion criteria were substance abuse or dependence within 6 months of screening, Diagnostic and Statistical Manual of Mental Disorders, 4th Edition, Text Revision (DSM-IV-TR) diagnosis of delirium or dementia, a score of ?17 on the minimental state examination, imminent risk of harm to self or others, or a score of 2 on items 7, 10, or 11 of the Modified InterSePT Scale for Suicidal Thinking (control over suicidal ideation, planning suicidal acts, expectation of committing suicide). Additional exclusion criteria were uncontrolled and unstable clinically significant medical conditions (e.g., renal, hepatic, endocrine, respiratory, cardiovascular, metabolic, hematologic, immunologic, or cerebrovascular disease; anorexia, obesity, or malignancy), jaundice or total bilirubin levels more than twice the upper limit of normal, narrow angle glaucoma, seizure disorder, serologic evidence of HIV infection, history of neuroleptic malignant syndrome, lifetime history of cerebrovascular accidents, myocardial infarction within the past 5 years, Parkinson's disease, treatment with clozapine in the preceding 6 months, participation in a previous asenapine clinical trial at any time or in any study of an investigational drug within the last 30 days, and judgement by the principal investigator that the patient would be unlikely to reduce daily benzodiazepine intake during the inpatient phase to a maximum of the equivalent of 6 mg/day of lorazepam. Although the same inclusion and exclusion criteria were used at each site, recruitment was not standardized with regard to ethnicity or other factors that might affect drug disposition.
Treatment
Participants were admitted to a hospital or long-term care facility 1 day before the first dose of trial medication and remained there through the first 8 treatment days. Consumption of grapefruit products 1 was prohibited because asenapine is a CYP3A4 substrate (2010b), although there is as yet no evidence that CYP 3A4 inhibitors and inducers do alter asenapine exposure to a degree that requires dose adjustment. After a washout period of up to 3 days, patients were randomized in a double-blind protocol (both patients and investigators were blind to the dosage regimen) to one of two dosing schedules. Group A patients received sublingual asenapine at 2 mg BID for the first 2 days, 5 mg BID for the next 2 days, and 10 mg BID thereafter. Group B patients received 5 mg BID for the first 4 days and 10 mg BID thereafter. If a patient failed to tolerate the 10-mg dose, the dose could be reduced to 5 mg after day 6. The maximum duration of exposure to asenapine 10 mg BID in patients completing 42 days of treatment was 38 days in both groups.
The titration schemes described earlier were developed based on multiple factors, including previous experience with asenapine, the objectives of the study, and the target population. A 2-day titration period was deemed sufficient because asenapine was generally well tolerated in early trials that used 1- to 3-day titration periods (unpublished data). A starting-dose period of 2 days (2 mg vs 5 mg) provided adequate opportunity to observe differences in adverse events (AEs) associated with the starting dose (especially orthostatic hypotension). The remainder of the titration schedule was designed to provide comparative data for PK analyses based on the estimated half-life of asenapine. The group B starting dose of 5 mg BID was chosen based on its demonstrated efficacy in the treatment of acute schizophrenia in younger adults (Potkin et al., 2007; Kane et al., 2010). The 2-mg BID dose for group A was chosen as a comparison based on the assumption that altered PK could make older patients less tolerant of any medication.
During the screening and inpatient periods, short half-life benzodiazepines (6 mg/day lorazepam or equivalent) were allowed for agitation and anxiety. Zolpidem, zaleplon, or zopiclone were permitted for insomnia. The use of anticholinergics or other EPS medications at the time of screening was permitted for the first 7 days after randomization. After day 7, their use was discontinued through a slow tapering process, but they could be reinstituted if EPS reemerged. The use of mood stabilizers and antidepressants, except CYP 1A2 inhibitors, could be continued throughout the trial provided that patients were stabilized on those drugs at the time of screening. Concomitant use of medications that interact with CYP enzymes that do not metabolize asenapine was permitted.
Pharmacokinetic assessments
Blood samples (5 mL) to measure the concentration of asenapine and its metabolite, N-desmethylasenapine, were collected in heparin-containing tubes before the morning dose on days 3, 4, 7, 8, 21, and 42 and sequentially at 0.5, 1, 2, 4, 8, and 12 h after the morning dose on days 4 and 8. Patients were instructed to record the administration times of the last 2 doses of trial medication before the collection of each blood sample during the outpatient phase.
Each collected sample was mixed by gentle inversion and placed into an ice-water bath. Plasma was isolated within approximately 30 min of collection by centrifugation (10-15min; 1000-1200Xg). The plasma was then collected, stored in a screw-capped plastic storage tube, frozen in an upright position at approximately —20°C within 60 min of sample collection, and stored on dry ice. Asenapine and N-desmethylasenapine concentrations were assessed using high-performance liquid chromatography coupled with mass spectrometric detection after solid phase extraction. The analytic range was 0.025-20.0 ng/mL for asenapine and 0.05-20.0 ng/ mL for N-desmethylasenapine.
In group A, noncompartmental PK analyses were performed after 4 days of exposure to 10 mg BID (cumulative days of asenapine exposure = 8); in group B, analyses were performed after 4 days of exposure to 5 mg BID (cumulative days of asenapine exposure = 4) and 4 days of exposure to 10 mg BID (cumulative days of asenapine exposure = 8). The following PK parameters were calculated for each patient based on the plasma samples collected on treatment days 4 and 8: peak concentration (Cmax), dose-normalized Cmax (dn-Cmax), tmax, area under the concentration versus time curve from 0-12 h (AUC0-12), dose-normalized AUC0-12 (dn-AUC0-12), and minimum predose concentration (Cmin). Cmax and tmax were taken from measured plasma concentrations; dn-Cmax was calculated as Cmax divided by the asenapine BID dose. AUC0-12 was calculated by means of the linear trapezoidal rule, with values below the lower limit of quantification substituted with 0; dn-AUC0-12 was then calculated as AUC0-12 divided by the asenapine BID dose.
Safety and tolerability assessments
Reported AEs were coded using the Medical Dictionary for Drug Regulatory Affairs (MedDRA version 10.1 [IFPMA, Chemin Louis-Dunant, Geneva, Switzerland]) and recorded throughout the study, as were their severity and relatedness to treatment. Vital signs were assessed at every visit, as was the use of concomitant medications. Clinical laboratory assessments (e.g., hematology, biochemistry, and urinalysis) and electrocardiograms were performed at screening and on treatment on days 8, 21, and 42/end of treatment (ET). Physical examinations were conducted at screening and on treatment day 42/ET. EPS were assessed through notation of spontaneously reported EPS-related AEs, as well as through a formal assessment using the abbreviated Extrapyramidal Symptom Rating Scale (ESRS-A) on treatment days 1, 8, 21, and 42/ET.
Psychotic symptom assessments
Psychotic symptoms were assessed by PANSS total and subscale scores, Marder factor scores, and CGI-S ratings. Assessments were conducted at screening and postdosing on treatment days 1, 8, 21, and 42/ET.
Statistical analyses
Safety was assessed in the treated population (all patients receiving ?1 dose of asenapine). PK analyses were conducted in the pharmacokinetically evaluable group (all patients who had ?1 PK parameter measured and no protocol violations that could alter PK [e.g., unscheduled dose titration or concomitant use ofnon-permitted medications]). Psychotic symptoms were assessed in the intent-to-treat (ITT) population (all treated patients who had ?1 postbaseline assessment of PANSS total score) using last observation carried forward (LOCF) to account for missing data. Descriptive summary statistics (e.g., mean, SD, and range) were used to describe safety, tolerability, PK, and psychotic symptoms.
Results
Disposition and demographics
Subject disposition is described in Figure 1. The F1 treated population comprised 122 patients (group A, n = 61; group B, n = 61; Figure 1); the most common reasons for not continuing in the 32 screened patients who did not enter the trial were not meeting the study criteria and withdrawn consent. The ITT population comprised 120 patients (group A, 59; group B, 61); 76 patients (group A, 36; group B, 40) completed the trial. The most common reasons for study discontinuation were AEs not related to worsening of symptoms, which were twice as common in group A versus group B, and withdrawn consent, which was comparable in groups A and B.
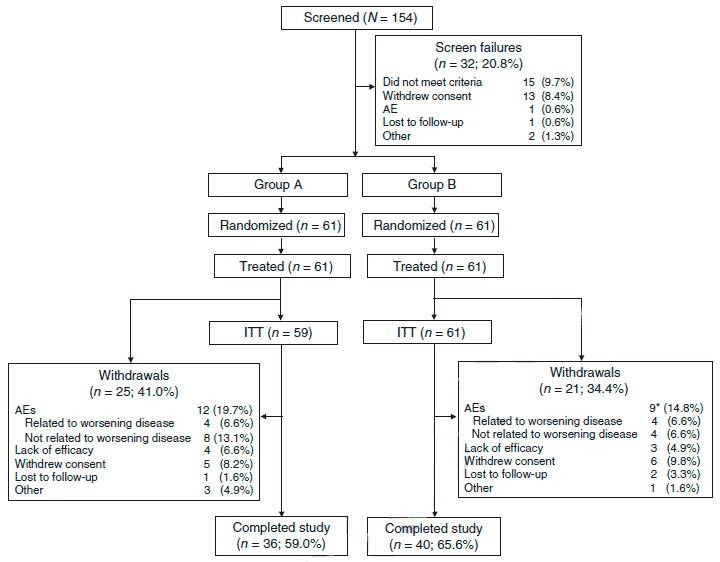
Figure 1 Patient disposition. AE, adverse event; ITT, intent to treat (treated patients with ?1 postbaseline assessment). *For one patient who discontinued owing to an AE, the relatedness of the AE to worsening of the disease was not available.
Most patients were White people (86.1%), women (72.1%), and ranged from 65 to 88 years of age; the most common psychiatric diagnoses were schizophrenia and major depressive disorder with psychotic features (Table 1). With few exceptions, baseline demographic T and clinical characteristics in the treated population were comparable between groups (Table 1). The proportion of women and patients older than 75 years was slightly higher in group B; the ethnic profile was similar between groups. Schizophrenia diagnoses were more common in group A, whereas schizoaffective disorder and psychotic disorders, not otherwise specified, were more common in group B.
The demographic characteristics of patients who discontinued treatment did not differ substantially from those of patients who completed the study. Of those patients who discontinued, 65.2% (30/46) were women and 80.4% (37/46) were White people; their mean ± SD age was 72.3 ± 5.9 years.
Extent of exposure and concomitant medications
Among all treated patients, the mean ± SD treatment duration was 31.7 ± 14.7 days (group A, 30.8 ± 15.3 days; group B, 32.7± 14.1 days). During the trial, including the initial dose-escalation period, the mean ± SD total daily asenapine dose was 14.6 ± 4.2 mg (group A, 14.1±4.7mg [95% CI, 12.9 to 15.3 mg]; group B, 15.2±3.6 mg [95% CI, 14.3 to 16.2 mg]).
Concomitant medications used by ?10% of patients in either treatment group were lorazepam, acetaminophen, zolpidem, docusate, lisinopril, aspirin, and valproate (Table 1). With the exception of zolpidem, use of these medications was 1.3- to 3.5-fold more common in group B than in group A; none of the medications used more frequently by group B are known to be inducers or inhibitors of the CYP3A4 isoenzyme.
Table 1
Demographics, clinical characteristics, and concomitant medication use (Treated population)
|
Characteristic |
Group A, n = 61 | Group B, n = 61 | All patients, n = 122 |
| Men, n (%) | 20 (32.8) | 14 (23.0) | 34 (27.9) |
| Women, n (%) | 41 (67.2) | 47 (77.0) | 88 (72.1) |
| Race, n (%) | |||
| White people | 52 (85.2) | 53 (86.9) | 105 (86.1) |
| Black people | 7 (11.5) | 7 (11.5) | 14 (11.5) |
| Asian* | 0 (0.0) | 1 (1.6) | 1 (0.8) |
| Other | 2 (3.3) | 0 (0.0) | 2 (1.6) |
| Mean age±SD, y | 70.5±4.6 | 72.0 ± 5.8 | 71.2 ±5.2 |
| 65-75, n (%) | 51 (83.6) | 44 (72.1) | 95 (77.9) |
| >75, n (%) | 10 (16.4) | 17 (27.9) | 27 (22.1) |
| Mean weight ±SD, kg | 70.2 ± 13.5 | 68.6±15.2 | 69.4 ± 14.3 |
| Mean BMI±SD, kg/m2 | 26.1 ±4.8 | 25.5 ± 5.1 | 25.8 ± 4.9 |
| Psychiatric diagnosis, n (%) | |||
| Major depressive disorder, severe with psychotic features | 8 (13.1) | 8 (13.1) | 16 (13.1) |
| Psychotic disorders, NOS | 2 (3.3) | 5 (8.2) | 7 (5.7) |
| Schizoaffective disorder | 3 (4.9) | 6 (9.8) | 9 (7.4) |
| Schizophrenia | 41 (67.2) | 30 (49.2) | 71 (58.2) |
| Paranoid type | 38 (62.3) | 28 (45.9) | 66 (54.1) |
| Residual type | 1 (1.6) | 2 (3.3) | 3 (2.5) |
| Undifferentiated type | 2 (3.3) | 0 (0.0) | 2 (1.6) |
| Mean MMSE score ± SD at screening | 25.2±3.4 | 24.8 ± 3.5 | 25.0 ± 3.5 |
| Concomitant medication use, n (%) | |||
| Any medication use | 50 (82.0) | 48 (78.7) | 98 (80.3) |
| Concomitant medication use by ?10% of patients | |||
| Lorazepam | 10 (16.4) | 16 (26.2) | 26 (21.3) |
| Acetaminophen | 8 (13.1) | 15 (24.6) | 23 (18.9) |
| Zolpidem | 10 (16.4) | 8 (13.1) | 18 (14.8) |
| Docusate | 6 (9.8) | 8 (13.1) | 14 (11.5) |
| Lisinopril | 5 (8.2) | 8 (13.1) | 13 (10.7) |
| Aspirin | 6 (9.8) | 7 (11.5) | 13 (10.7) |
| Valproate | 2 (3.3) | 7 (11.5) | 9 (7.4) |
Pharmacokinetics
Asenapine and N- desmethylasenapine PK parameters are presented in Table 2. Compared with adult schizophrenia or patients with schizoaffective disorder 18 to 65 years exposed to 5 mg BID asenapine for 10 days and 10 mg BID asenapine for 5 days (Chapel et al., 2009), asenapine Cmax (geometric means) in this older population were 12% higher with 5 mg BID (on study day 4) and 21% higher with 10 mg BID (on study day 8); AUC0-12 values were 21% higher with 5 mg BID and 30% higher with 10 mg BID. Exposure to the largely inactive metabolite N-desmethylasenapine in older patients versus 18- to 65-year-old patients (data on file) was 75% higher for Cmax with 5 mg BID and 63% higher for Cmax at 10 mg BID; AUC0-12 values were 77% higher with 5 mg BID and 72% higher with 10 mg BID.
Table 2
Summary of pharmacokinetic parameters* for asenapine and N-desmethylasenapine (All pharmacokinetically evaluable patients)
|
Patients ?65y (this study) |
Patients 18-65 y† | |||||||
|
Asenapine 5mg BID |
Asenapine 10 mg BID |
Asenapine 5 mg BID |
Asenapine 10mg BID | |||||
| n | Data | n | Data | n | Data | n | Data | |
| Asenapine tmax, h | 87 | 1.00 (0.42-4.23) | 60 | 1.06 (0.42-4.08) | - | - | - | - |
| Cmax, ng/mL | 87 | 4.73 (89.5) | 60 | 7.93 (95.3) | 28 | 4.23 (45) | 25 | 6.56 (51) |
| dn-Cmax, ng/mL/mg | 87 | 0.946 (89.5) | 60 | 0.793 (95.3) | - | - | - | - |
| AUC0-12, ng-h/mL | 87 | 32.1 (76.0) | 60 | 56.3 (84.9) | 28 | 26.5 (38) | 25 | 43.4 (53) |
| dn-AUC0-12, ng-h/mL/mg | 87 | 6.42 (76.0) | 60 | 5.63 (84.9) | - | - | - | - |
| C min | 86 | 1.74 (90.9) | 60 | 3.25 (79.4) | - | - | - | - |
| N- desmethylasenapine t max, h | 85 | 4.00 (0.50-12.0) | 60 | 4.00 (0.50-12.0) | - | - | - | - |
| Cmax, ng/mL | 85 | 1.78 (51.9) | 60 | 3.61 (51.1) | 28 | 1.02 (39) | 25 | 2.21 (48) |
| dn-Cmax, ng/mL/mg | 85 | 0.356 (51.9) | 60 | 0.361 (51.1) | - | - | - | - |
| AUC0-12, ng-h/mL | 85 | 17.1 (50.6) | 60 | 33.8 (55.5) | 28 | 9.65 (39) | 28 | 19.6 (36) |
| dn-AUC0-12, ng-h/mL/mg | 85 | 3.41 (50.6) | 60 | 3.38 (55.5) | - | - | - | - |
| Cmin | 84 | 1.05 (55.1) | 60 | 2.34 (58.0) | - | - | - | - |
Safety and tolerability
Adverse events. The overall incidence of treatment-emergent AEs among all treated patients was comparable in each treatment group (Table 3). Most AEs were mild or moderate in intensity. The incidence of mild intensity AEs was higher in group B; the incidence of moderate and severe intensity AEs, as well as the incidence of serious AEs, was higher in group A. The overall incidence ofdiscontinuation because ofAEs was 17.2% and was slightly higher in group A. A 76-year-old patient died because of cardiorespiratory arrest 29 days after the last dose of trial medication (the patient had received medication for 42 days); this death was considered by the investigator to be unrelated to treatment.
Table 3
Summary of adverse events (Treated population)
| Number (%) of patients | |||
| Group A, n = 61 | Group B, n = 61 | All patients N =122 | |
| All treatment-emergent AEs/serious AEs | 44 (72.1) | 44 (72.1) | 88 (72.1) |
| Mild | 10 (16.4) | 20 (32.8) | 30 (24.6) |
| Moderate | 25 (41.0) | 20 (32.8) | 45 (36.9) |
| Severe | 9 (14.8) | 4 (6.6) | 13 (10.7) |
| All treatment-emergent serious AEs | 6 (9.8) | 3 (4.9) | 9 (7.4) |
| DC caused by treatment-emergent AEs/serious AEs Treatment-emergent AEs in ?5% of patients | 12 (19.7) | 9 (14.8) | 21 (17.2) |
| Hypertension | 7 (11.5) | 3 (4.9) | 10 (8.2) |
| Headache | 4 (6.6) | 4 (6.6) | 8 (6.6) |
| Somnolence | 5 (8.2) | 3 (4.9) | 8 (6.6) |
| Asthenia | 4 (6.6) | 2 (3.3) | 6 (4.9) |
| Dizziness | 4 (6.6) | 2 (3.3) | 6 (4.9) |
| Parkinsonism | 1 (1.6) | 5 (8.2) | 6 (4.9) |
| Blood pressure increase | 0 (0.0) | 5 (8.2) | 5 (4.1) |
| Anxiety | 0 (0.0) | 4 (6.6) | 4 (3.3) |
| Urinary tract infection | 4 (6.6) | 0 (0.0) | 4 (3.3) |
Treatment-emergent AEs occurring in ?5% of all patients included hypertension, headache, and somnolence. Hypertension, somnolence, headache, asthenia, dizziness, and urinary tract infection occurred in ?5% of patients in group A and Parkinsonism, headache, blood pressure increase, and anxiety occurred in ?5% of patients in group B (Table 3). The incidence of treatment-emergent AEs during the first 2 days of treatment was assessed to compare the 2 mg BID and 5 mg BID dosages. During the first 2 days, the incidence of somnolence was 3.3% (2/61) in each treatment group, the incidence of orthostatic hypotension was 3.3% (2/61) in group Aand 1.6% (1/61) in group B; the incidence of blood pressure increase was 3.3% (2/61) in group B and 0% in group A.
Extrapyramidal symptoms. The overall incidence of treatment-emergent EPS in all treated patients was 5.7% (group A, 3.3%; group B, 8.2%; p = 0.2448 based on post hoc analysis with a Cochran-Mantel-Haenszel chi-square test). In group A, one case each of Parkinsonism and akathisia were reported. In group B, Parkinsonism accounted for all five cases of EPS-related AEs. Although most EPS-related AEs were reported at 10 mg BID (group A, n = 1/2 [50%]; group B, n = 4/5 [80%]), the number of patients is too small to draw conclusions regarding possible dose effects.
The mean ± SD baseline ESRS-A score was 5.4 ± 7.8 (group A, 5.6±8.4; group B, 5.1 ±7.1). Mean±SD changes from baseline in ESRS-A total score in all patients were -1.0 ± 5.8 at day 8 (group A, -1.4 ±4.1; group B, -0.5 ±7.1), -1.1 ± 5.2 at day 21 (group A, -0.6 ±4.6; group B, -1.6 ±5.7), and -1.6 ± 5.2 at day 42 (group A, -0.8 ±5.0; group B, -2.2 ±5.4).
Other safety and tolerability measures. Table 4 summarizes changes in anthropometric measures, body mass index (BMI), and metabolic syndrome in all treated patients. Overall changes from baseline in weight and abdominal girth during the study were minimal. There was tendency for weight and abdominal girth to increase more in group B; however, the number of patients and the magnitude of the difference preclude any inference of the significance of the difference. Although most patients remained in the same body BMI category from baseline to study end, shifts to the next higher BMI category appeared to be more common in group B, and shifts to the next lower BMI category appeared to be more common in group A. At baseline, the percentage of patients meeting the National Cholesterol Education Program criteria for the metabolic syndrome was comparable across treatment groups; the percentage meeting these National Cholesterol Education Program criteria decreased in group A and was unchanged in group B at study end.
Table 4
Summary of anthropometric measures, BMI shifts, and metabolic syndrome (Treated population)
| Group A | Group B | All patients | |
| Clinically significant weight gain*, n/N (%) | 0(0) | 2 (3.3) | 2 (1.6) |
| Weight change from baseline, kg | |||
| Mean ± SD at baseline | 70.2±13.5 | 68.6 ± 15.2 | 69.4 ± 14.3 |
| Mean ± SD change from baseline | 0.1 ± 2.4 | 0.8±2.4 | 0.4±2.4 |
| Abdominal girth, cm | |||
| Mean ± SD at baseline | 89.1 ± 14.5 | 91.4 ± 16.6 | 90.3 ± 15.6 |
| Mean ± SD change from baseline | 0.5 ± 5.2 | 3.5 ± 17.0 | 2.0 ± 12.7 |
| BMI | |||
| Shift to next higher category, n/N (%) | 2/39 (5.1) | 7/43 (16.3) | 9/82 (11.0) |
| Shift to next lower category, n/N (%) | 3/39 (7.7) | 1/43 (2.3) | 4/82 (4.9) |
| Metabolic syndrome | |||
| Met NCEP criteria at baseline, n/N (%) | 5/30 (16.7) | 6/37 (16.2) | 11/67 (16.4) |
| Met NCEP criteria and endpoint, n/N (%) | 3/30 (10.0) | 6/37 (16.2) | 9/67 (13.4) |
| Met NCEP criteria at end point but not baseline, n/N (%) | 2/30 (6.7) | 2/37 (5.4) | 4/67 (6.0) |
| Met NCEP criteria at baseline but not end point, n/N (%) | 4/30 (13.3) | 2/37 (5.4) | 6/67 (9.0) |
*Clinically significant weight gain (?7% increase from baseline).
Prolactin levels fell in both groups. Insulin levels rose in group B only, an effect that is attributed to a single patient whose insulin level increased to 500 pmol/L over baseline. Changes, deemed clinically significant by the investigators, were noted in the electrocardiograms of seven (5.7%) patients (group A, n = 5 [8.2%]; group B, n = 2 [3.3%]). Of these, three were cases of sinus bradycardia and one each were cases of sinus tachycardia, atrial or supraventricular tachycardia, atrial fibrillation, and QT prolongation. Among all patients, the mean ± SD QTc interval remained essentially unchanged (baseline, 418.6 ± 47.6 ms; endpoint, 417.4 ± 27.0 ms).
Psychotic symptoms. Psychotic symptoms in the ITT population (LOCF analysis) decreased slightly on average during the study period (Table 5), with no evidence of worsening symptoms.
Table 5
Change from baseline to study endpoint (Mean ± SD) for psychotic symptom measures (ITT population, LOCF)*
| Group A (n = 59) | Group B (n = 61) | All patients (N = 120) | ||||
| Baseline | Change at end point | Baseline | Change at end point | Baseline | Change at end point | |
| PANSS scores | ||||||
| Total | 84.5 ± 15.0 | -10.6 ± 16.7 | 84.8 ± 14.8 | -18.2 ± 15.9 | 84.7 ± 14.9 | -14.5 ± 16.7 |
| Positive subscale | 21.8 ± 4.4 | -4.9 ± 5.7 | 21.6 ± 4.4 | -6.3 ± 5.8 | 21.7 ± 4.4 | -5.6 ± 5.7 |
| Negative subscale | 21.6 ± 6.8 | -1.3 ± 5.1 | 20.2 ± 6.5 | -3.3 ± 4.4 | 20.9 ± 6.6 | -2.3 ± 4.8 |
| Global psychopathology subscale | 41.0 ± 8.1 | -4.5 ± 9.0 | 43.0 ± 8.5 | -8.6 ± 8.6 | 42.0 ± 8.3 | -6.6 ± 9.0 |
| PANSS Marder factor symptom scores | ||||||
| Positive | 25.7 ± 5.4 | -4.8 ± 5.6 | 25.5 ± 4.8 | -6.3 ± 5.6 | 25.6 ± 5.1 | -5.6 ± 5.7 |
| Negative | 20.1 ± 6.5 | -1.5 ± 5.7 | 18.6 ± 7.0 | -3.0 ± 5.0 | 19.3 ± 6.8 | -2.3 ± 5.4 |
| Disorganized thought | 20.2 ± 5.1 | -1.4 ± 3.8 | 19.9 ± 4.6 | -3.8±3.9 | 20.0 ± 4.8 | -2.6 ± 4.0 |
| Hostility/excitement | 9.4 ± 3.5 | -1.4 ± 3.7 | 10.0 ± 3.7 | -2.5 ± 3.7 | 9.7 ± 3.6 | -2.0 ± 3.7 |
| Anxiety/depression | 9.3 ± 3.1 | -1.5 ± 2.9 | 10.8 ± 3.9 | -2.6 ± 3.3 | 10.1 ± 3.6 | -2.1 ± 3.2 |
| CGI-S score | 4.3 ± 0.8 | -0.6 ± 1.1 | 4.4 ± 0.6 | -0.9 ± 1.0 | 4.3 ± 0.7 | -0.8 ± 1.1 |
Discussion
Asenapine exposure was higher in older patients with psychosis than was previously reported in adults 18 to 65 years with schizophrenia (Chapel et al., 2009), suggesting a decreased drug clearance in older patients. In adults 18 to 65 years with schizophrenia or schizoaffective disorder, 10 days of asenapine 5 mg BID produced a mean steady-state Cmax of 4.2 ng/mL, mean tmax of 1.8 h, and AUC0-12 of 26.5 ngh/mL; after 5 days of asenapine 10 mg BID, mean steady-state Cmax was 6.6 ng/mL, tmax was 2.0 h, and AUC0-12 was 43.4 ngh/mL (Chapel et al., 2009). Compared with these asenapine exposure levels (Chapel et al., 2009), asenapine Cmax was 12 to 21% higher and AUC0-12 was 21 to 30% higher in these older patients. Exposure to the largely inactive metabolite N-desmethylasenapine in this older population was up to 77% higher compared with adult patients ?65 years (data on file).
Differences between the Chapel et al. (2009) study and the current study need to be considered when comparing asenapine exposure between older and younger age groups. First, in the current study, steady-state asenapine exposure was assessed in patients who received 5 or 10 mg BID asenapine for 4 consecutive days (cumulative asenapine exposure ranged from 4 to 8 days). In the Chapel et al. study (2009), assessments were made after 10 and 5 consecutive days of exposure, respectively, to 5 and 10 mg BID asenapine (cumulative exposure: 10 and 16 days, respectively). Because steady-state asenapine levels are usually attained within 4 - 6 days of exposure in younger patients (Dogterom et al., 2009), these differences in exposure duration prior to PK measurements may not have substantially contributed to the age differences in he pharmacokinetic profile of asenapine. However, time to steady state may be longer in older patients. Another issue is that there were differences in the ethnic distributions between studies (Chapel et al. (2009): White people [32%], Black people [50%], other [18%]; Current study: White people [86%], Black people [12%], other [2%]), possibly leading to differences in CYP450 polymorphisms among patient groups. CYP1A2 is the predominant CYP450 isoenzyme responsible for the oxidative metabolism of asenapine (2010b). As the activity of CYP1A2 has been reported to be lower in Black people compared with White people (Relling et al., 1992), the higher proportion of Black people in the Chapel study would be expected to minimize, not accentuate, PK differences between the studies. Therefore, ethnic differences may not have contributed to the increased exposure observed in the older population assessed in he current study, although the possibility that differences in CYP1A2 activity existed between the diverse groups of White patients in the two studies cannot be excluded.
Taken together, these observations suggest that greater asenapine exposure occurs in older patients than in younger patients. Increased exposure to asenapine in older patients may be a result of decreased hepatic blood flow, which is the main determinant of asenapine clearance (2010b), with ncreases in N-desmethylasenapine exposure being more pronounced than asenapine because it has fewer alternative metabolic pathways. Altered absorption seems unlikely because drug absorption is decreased in older patients, and increased adipose issue in older patients would be expected to produce a larger volume of distribution and lower levels. Nevertheless, asenapine appeared to be well tolerated n this population and rapid dose escalation seems ustified.
Interpretation of the results of this study is limited by the small number of patients and the short duration of the study, both of which precluded meaningful assessment of the risk of long-term adverse effects such as cerebrovascular accidents, sudden death, weight gain, and metabolic syndrome. The lack of placebo control after the brief dosage escalation and the lack of formal statistical assessment of safety parameters also limit interpretation of these data. However, with a few exceptions, the incidence of AEs was comparable across groups, as was the overall safety and tolerability profile. Although the incidence of moderate to severe intensity AEs and of orthostatic hypotension at day2 was numerically higher in group A,which could have contributed to the higher incidence of discontinuation owing to AEs in group A, the small sample size makes it impossible to make statistical inferences about the apparent between-group differences, including differences in discontinuation rates. For example, only four more patients in group A than in group B developed hypertension, a difference that is not likely to be meaningful. Although EPS-related AEs occurred at asenapine 10 mg BID in most patients, the risk of EPS with 10 mg BID should be considered cautiously because from day8 onward (when all patients were taking 10 mg BID asenapine) mean ESRS-A scores were decreased compared with baseline.
Although a direct comparison has not been conducted, the types of AEs reported in this older population appear to be comparable with those observed in younger patients with schizophrenia (Potkin et al., 2007; Kane et al., 2010) and in older patients treated with other atypical antipsychotics (Sajatovic et al., 1998; Tariot et al., 2000; Barak et al., 2002; Lasser et al., 2004; Tzimos et al., 2008; Riedel et al., 2009). In the older patients who participated in this study, the incidence of common AEs, such as headache and somnolence, was about the same as the incidence previously reported in younger patients with schizophrenia treated with asenapine for up to 6 weeks (Potkin et al., 2007; Kane et al., 2010) and younger patients with bipolar mania treated for up to 12 weeks (McIntyre et al., 2009a; McIntyre et al., 2009b; McIntyre et al., 2010). However, it should also be noted that, although the types of AEs and the overall incidence of treatment-emergent AEs reported in this older population are similar to those reported in trials that had younger populations, discontinuations attributed to AEs was higher in this older population than in younger patients with schizophrenia (17.2 vs 5-10%) (Potkin et al., 2007; Kane et al., 2010), perhaps suggesting that this older population may have found common AEs to be less tolerable.
Although patients in this study exhibited decreased levels of psychotic symptoms as measured by the PANSS, it is not possible to make a definitive assessment of the efficacy of asenapine for treating psychotic symptoms in this older population. The design of the study, including the absence of a placebo group, the lack of a formal DSM diagnosis for every subject (only predefined psychotic symptoms), the open label prescribing method (i.e., all patients received active drug), and the liberal use of concomitant medications make it difficult to interpret measures of efficacy, especially because the 17% decrease in PANSS scores is lower than the customary 30% defined as response in schizophrenia studies. Because this study was not powered for an analysis of efficacy, inferences about efficacy are not possible.
Conclusion
Sublingual asenapine at a dosage of 5 or 10 mg BID appeared to be as well tolerated in patients ?65 years with psychosis, despite the higher exposure levels observed in comparison with younger patients with schizophrenia. Although the numbers were too small to permit statistical analysis of group differences, asenapine tolerability was generally comparable over two dose-escalation regimens, and the tolerability and safety profiles were comparable with those of younger adults. In light of these findings, no dosage change is anticipated to be necessary in older adults with nondementia-related psychosis, and the simplest titration method can be used. Because substantial differences in the safety/tolerability of asenapine were not observed between the two dose-escalation regimens, rapid titration from 5 mg BID to 10 mg BID seems acceptable. Limitations of the method preclude comments about the effectiveness of asenapine in older psychotic patients.
Conflicts of interest
Dr Dubovsky has received research support from Pfizer, Otsuka, Sumitomo Pharma, Biogen, Merck, and the Tower Foundation. Drs Frobose and Phiri are employees of Merck. Dr de Greef is an employee of Merck Sharp & Dohme. Dr Panagides was an employee of Schering-Plough (formerly Organon), now Merck, at the time this study was conducted.
Acknowledgements
Financial support for this study was provided by Merck (Whitehouse Station, NJ). Editorial assistance was provided by Complete Healthcare Communications, Inc. (Chadds Ford, PA) with funding from Merck.
Key points
Asenapine exposure was higher in older patients with psychosis than previously reported in younger adults with schizophrenia, suggesting decreased drug clearance in older patients.
Asenapine appeared to be well tolerated in this population.
REFERENCES
European Medicines Agency. Sycrest (asenapine).
Saphris® (asenapine sublingual tablets). Full Prescribing Information, Schering Corporation, a subsidiary of Merck & Co., Inc, Whitehouse Station, NJ, 2010b.
Barak Y, Shamir E, Zemishlani H, et al. 2002. Olanzapine vs. haloperidol in the treatment of elderly chronic schizophrenia patients. Prog Neuropsychopharmacol Biol Psychiatry 26: 1199-1202.
Chapel S, Hutmacher MM, Haig G, et al. 2009. Exposure-response analysis in patients with schizophrenia to assess the effect of asenapine on QTc prolongation. J Clin Pharmacol 49: 1297-1308.
Dogterom P, Timmer C, deGreef R, et al. A phase I study to investigate the safety, tolerability, and pharmacokinetics of single and multiple doses of sublingually administered asenapine in healthy male volunteers. Presented at: American Society for Clinical Pharmacology and Therapeutics 110th Annual Meeting; 3-8 May 2009; Washington, DC.
Douglas IJ, Smeeth L. 2008. Exposure to antipsychotics and risk of stroke: self controlled case series study. BMJ 337: a1227.
Gareri P, De Fazio P, De Fazio S, et al. 2006. Adverse effects of atypical antipsychotics in the elderly: a review. Drugs Aging 23: 937-956.
Gill SS, Rochon PA, Herrmann N, et al. 2005. Atypical antipsychotic drugs and risk of ischaemic stroke: population based retrospective cohort study. BMJ 330: 445.
Herrmann N, Mamdani M, Lanctot KL. 2004. Atypical antipsychotics and risk of cerebrovascular accidents. Am J Psychiatry 161: 1113-1115.
Hybels CF, Blazer DG. 2003. Epidemiology of late-life mental disorders. Clin Geriatr Med 19: 663-696.
Jeste DV, Alexopoulos GS, Bartels SJ, et al. 1999. Consensus statement on the upcoming crisis in geriatric mental health: research agenda for the next 2 decades. Arch Gen Psychiatry 56: 848-853.
Kane JM, Cohen M, Zhao J, Alphs L, Panagides J. 2010. Efficacy and safety of asenapine in a placebo—and haloperidol—controlled trial in patients with acute exacerbation of schizophrenia. J Clin Psychopharmacol 30: 106-115.
Klotz U. 2009. Pharmacokinetics and drug metabolism in the elderly. Drug Metab Rev 41: 67-76.
Lasser RA, Bossie CA, Zhu Y, et al. 2004. Efficacy and safety of long-acting risperidone in elderly patients with schizophrenia and schizoaffective disorder. Int J Geriatr Psychiatry 19: 898-905.
McIntyre RS, Cohen M, Zhao J, et al. 2009a. A 3-week, randomized, placebo-controlled trial of asenapine in the treatment of acute mania in bipolar mania and mixed states. Bipolar Disord 11: 673-686.
McIntyre RS, Cohen M, Zhao J, et al. 2009b. Asenapine versus olanzapine in acute mania: a double-blind extension study. Bipolar Disord 11: 815-826.
McIntyre RS, Cohen M, Zhao J, et al. 2010. Asenapine in the treatment of acute mania in bipolar I disorder: a randomized, double-blind, placebo-controlled trial. J Affect Disord 122: 27-38.
Potkin SG, Cohen M, Panagides J. 2007. Efficacy and tolerability of asenapine in acute schizophrenia: a placebo- and risperidone-controlled trial. J Clin Psychiatry 68: 1492-1500.
Prohn M, de Greef HJMM, Chapel S, Kerbusch T. 2009. Population pharmacokinetics of asenapine in patients with schizophrenia or bipolar disorder Eur Neuropsychopharmacol 19: S542-S543.
Rapoport M, Mamdani M, Shulman KI, Herrmann N, Rochon PA. 2005. Antipsychotic use in the elderly: shifting trends and increasing costs. Int J Geriatr Psychiatry 20: 749-753.
Regier DA, Boyd JH, Burke JD Jr, et al. 1988. One-month prevalence of mental disorders in the United States. Based on five epidemiologic catchment area sites. Arch Gen Psychiatry 45: 977-986.
Relling MV, Lin JS Ayers GD, Evans WE. 1992. Racial and gender differences in N-acetyltransferase, xanthine oxidase, and CYP1A2 activities. Clin Pharmacol Ther 52: 643-658.
Riedel M, Eich FX, Moller HJ. 2009. A pilot study of the safety and efficacy of amisulpride and risperidone in elderly psychotic patients. Eur Psychiatry 24: 149-153.
Sajatovic M, Perez D, Brescan D, Ramirez LF. 1998. Olanzapine therapy in elderly patients with schizophrenia. Psychopharmacol Bull 34: 819-823.
Schoemaker J, Naber D, Vrijland P, Panagides J, Emsley R. 2010. Long-term assessment of asenapine vs. olanzapine in patients with schizophrenia or schizoaffective disorder. Pharmacopsychiatry 43: 138-146.
Tariot PN, Salzman C, Yeung PP, Pultz J, Rak IW. 2000. Long-term use of quetiapine in elderly patients with psychotic disorders. Clin Ther 22: 1068-1084.
Tzimos A, Samokhvalov V, Kramer M, et al. 2008. Safety and tolerability of oral paliperidone extended-release tablets in elderly patients with schizophrenia: a double-blind, placebo-controlled study with six-month open-label extension. Am J Geriatr Psychiatry 16: 31-43.
Uchida H, Mamo DC, Mulsant BH, Pollock BG, Kapur S. 2009. Increased antipsychotic sensitivity in elderly patients: evidence and mechanisms. J Clin Psychiatry 70: 397-405.
