Long-term Opioid Blockade and Hedonic Response: Preliminary Data from Two Open-Label Extension
СтатьиSponsored by Alkermes, Inc., Cambridge, MA, and Cephalon, Inc., and Frazer, PA.
Poster presented at the American Society of Addiction Medicine, April 11-13, 2008, Toronto
Studies with Extended-Release Naltrexone
Charles O'Brien, MD, PhD1, David R. Gastfriend, MD2, Robert F. Forman, PhD2, Helen M. Pettinati, PhD1
1Department of Psychiatry, University of Pennsylvania School of Medicine, Philadelphia, PA; 2Alkermes, Inc., Cambridge MA
ABSTRACT
Background: Monthly injection of extended-release naltrexone (XR-NTX) in conjunction with psychosocial support is an effective treatment for alcohol dependence. Given that this treatment may be sustained over long durations, we examinedits long-term effects on hedonic response.
Method: This analysis examined hedonic response following long-term (3 to 5 years) continuous treatment with XR-NTX in two separate open-label extension studies. Patients rated degree of pleasurable response to 13 activities during the past 90 days using a 5-point Likert-like scale anchored with 1 ("not at all") to 5 ("very much so").
Results: For 74 alcohol dependent patients with a #.# year mean duration of XR-NTX, the mean level of hedonic response to drinking alcohol was significantly lower than the mean level of hedonic response to a wide range of other activities. The lowest mean pleasurable ratings were obtained for gambling (2.46), drinking alcohol (2.62), and shopping (2.67). These categories were quantitatively and qualitatively distinct, both in terms of median scale ratings and effect size of hedonic response, from others, including eating, sex, and exercise. Eating good food (4.00), listening to music (3.94), being with friends (3.63), and sexual relations (3.44) were among the highest mean ratings.
Conclusion: Long-term treatment with XR-NTX appears compatible with hedonic responsivity to a wide range of activities including eating good food, listening to music, being with friends and sex. Relative to these activities, however, its effect appears to diminish pleasure from gambling, drinking alcohol, and shopping. These findings have important implications for clinical practice and future research.
INTRODUCTION
• An FDA-approved injectable formulation of naltrexone (XR-NTX; Vivitrol®) provides continuous release of naltrexone for approximately 30 days following each injection.
• Naltrexone is an opioid antagonist that is thought to block the reinforcing effects of alcohol (King et al., 1997).
• The extent to which extended-release naltrexonemay block the reinforcing effects of other pleasurable activities is unknown.
OBJECTIVE
To determine whether long-term XR-NTX treatment impacts patients' experience of pleasure from a variety of everyday activities
METHODS
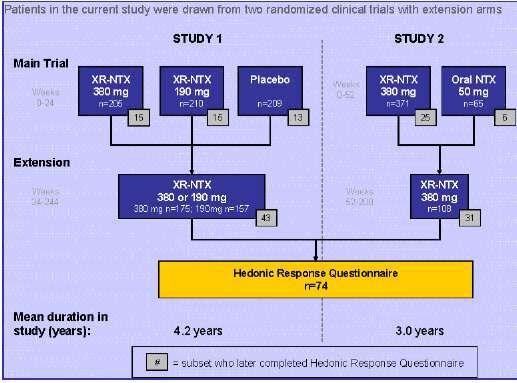
Subjects: Subjects had participated in one of two randomized clinical trials (Garbuttet al., 2005; Alkermes, Inc., Data on File) evaluating the efficacyand safety of XR-NTX as a treatment for alcohol dependence. Following 6-month (Study 1) or 1-year (Study 2) initial randomized trials, patients were eligible to participate in extension phases lasting 3-5 years (Figure 1).
Key Inclusion Criteria for Initial Randomized Trials
• DSM-IV diagnosis of Alcohol dependence
• Adult outpatients, ages 18 and older
• Minimum of 2 episodes of heavy drinking (> 5 standard drinks/dayfor men; > 4 for women) per week the 30 days before screening.
• Signed written, informed consent; study approved by ethics review board
Figure 1. Patient Flow
Key Exclusion Criteria for Initial Randomized Trials
• Untreated major depression with suicidal ideation; psychosis, or bipolar disorder
• Dependence within the past year on benzodiazepines, opiates, or cocaine
Procedures
• After the extension phases of the clinical trials, subjects who continued on XR-NTX were invited to participate in the current questionnaire study.
Treatment
• XR-NTX was given every 4 weeks as an intramuscular glutealinjection, alternating sides with each visit.
• At the time of participation in the current study, 53 (71%) of patients were receiving 380 mg of XR-NTX and 21 (29%) were receiving 190 mg of XR-NTX.
• Subjects in Study 1 and Study 2 had received XR-NTX for an average of 4.2 years and 3.0 years, respectively, or #.# years, overall.
• Adherence with monthly injections of XR-NTX over these extended time periods averaged 99.8% for Study 1 and 99.7% for Study 2.
Hedonic Response Questionnaire
• We created a 13-item hedonic response questionnaire based on pleasurable activities that might be affected by opioid antagonists. From the literature on the effects of opioid antagonists, items related to alcohol (King et al., 1997), physical exercise (Daniel et al., 1992; Jarvekulg et al., 2002), gambling (Kim et al., 2001), eating (Yeomans & Gray, 2002), sex (Murphy et al., 1990), and shopping (Kim, 1998) were created. Additional activities included: listening to music, reading, being with friends, playing video/card games, and watching sports.
• For each activity they had engaged in within the past 90 days, respondents rated how pleasurable the activity was on a 1 ("not at all") to 5 ("very much so") scale.
Statistical Analyses
• We tested the hypothesis that XR-NTX selectively limits hedonic response to alcohol vs. other pleasurable activities. The overall mean ratings for hedonic response to drinking alcohol vs.each of the other activities were compared using paired t-tests. Effect sizes were calculated using Cohen's d (difference between means divided by the pooled standard deviation).
RESULTS
Baseline Characteristics
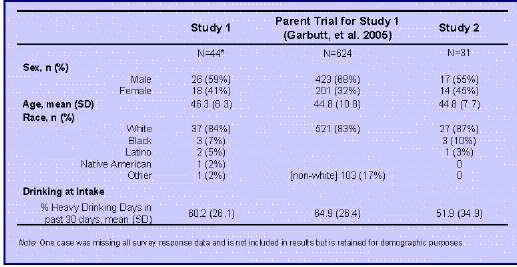
• Demographic characteristics of the sample and mean severity of drinking at baseline (before beginning XR-NTX) are shown in Table 1.Baseline characteristics of these 74 patients were similar to those of the full sample of patients who originally began XR-NTX in the parent studies.
Table 1. Clinical and Demographic Characteristics of Sample
Pleasure from Drinking Alcohol
• At this long-term assessment point, patients who had consumed alcohol during the past 90 days showed relatively low ratings of pleasure derived from drinking (mean = 2.57 on 1 to 5 scale) with only 19% rating "drinking alcohol" as "very much" or "quite a bit" pleasurable.
Pleasure from Everyday Activities
• Most of the everyday activities, including eating good food, sex, and exercise, were rated as moderately pleasurable or better by a majority of respondents, with the exception of: drinking alcohol, gambling, and shopping (Figure 2). The top-rated activities were:
• 93% of subjects rated listening to music " as "very much," "quite a bit" or "moderately" pleasurable.
• 89% of subjects rated "eating good food" as "very much," "quite a bit" or "moderately" pleasurable.
• 87% of subjects rated "being with friends" as "very much," "quite a bit" or "moderately" pleasurable.
• 60% of subjects rated "exercising or participating in sports" as "very much," "quite a bit" or "moderately" pleasurable.
Pleasurable Everyday Activities in Comparison to Drinking Alcohol
• Compared to drinking alcohol, mean pleasure ratings were significantly higher for most other activities: listening to music, sex, reading, being with friends, eating good food, eating spicy food, and playing video/card games (all p's < 0.005) (Table 2).
• Among patients who gambled in the previous 90 days (n = 26), hedonic response to this activity was not significantly different (p= 0.096) from hedonic response to drinking alcohol.
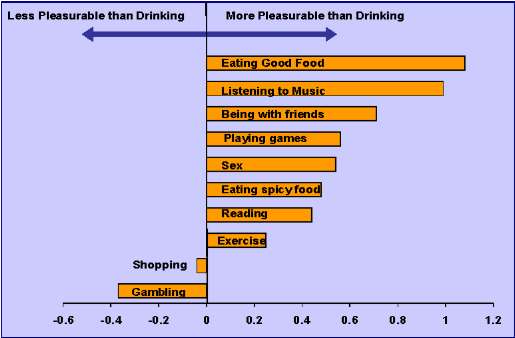
A qualitative distinction was found in effect sizes comparing everyday activities to drinking alcohol in which these were positive for a wide range of activities including eating good food, sex and to exercise, in contrast to negative effect sizes for gambling and shopping (Figure 3). Findings did not vary significantly by dosage of XR-NTX (190 mg vs. 380 mg) or years of exposure to XR-NTX.
Figure 2:Pleasure from Everyday Activities: Percent of Patients Endorsing Each Response
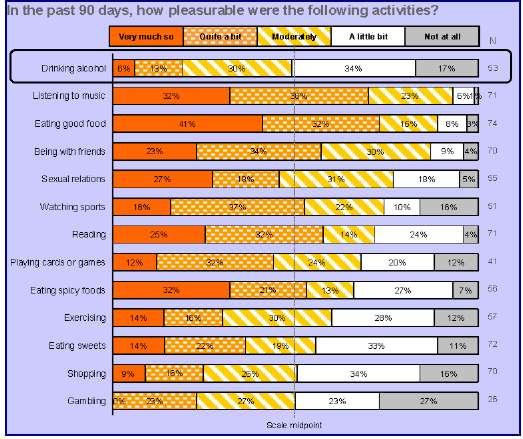
Table 2. Mean Hedonic Response to Pleasurable Activities Following 3-5 Years of XR-NTX Treatment
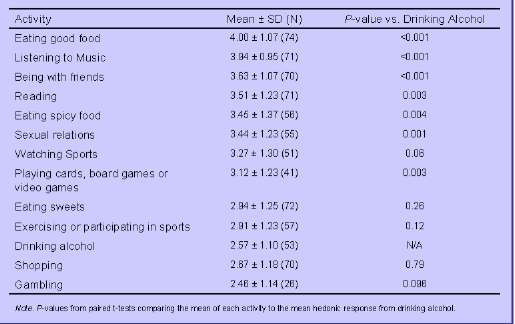
Figure 3: Effect Sizes Comparing Ratings of Pleasure from Everyday Acivities to Pleasure from Drinking Alcohol
CONCLUSIONS
• This is the first studyto assess hedonic response during assured, continuous, long-term administration ofa mu-opioidreceptorantagonist.
• Limitations ofthis exploratoryinvestigation include the absence ofa baseline hedonic effect measurement and placebo control, a small and self-selectedsampleanduse ofa previouslynon-validatedmeasure. Strengths includethelong durationofuse, theinternalvaliditysuggestedby contrasting responses to differentactivities, and the consistencyoffindings regardless of dose.
• Long-term treatment with extended-release naltrexone appears to selectively inhibit the hedonic response associated with drinking alcohol, while sparing the experience of pleasure associated with other activities such as listening to music, being with friends, sex, eating good food, and reading.
• Of 13 items studied, gambling, drinking alcohol and shopping received the lowest mean ratings of hedonic experience among patient treated with extended-release naltrexone for 3-5 years.
• The data indicate that clinical use of extended-release naltrexone by alcohol dependent patients is compatible with pleasurable response to a variety of everyday activities while inhibiting the pleasure associated with drinking alcohol.
REFERENCES
Daniel M, Martin AD, Carter J. Opiate receptor blockade by naltrexone and mood state after acute physical activity. Br J Sports Med.1992;26:111-115.
Garbutt JC, Kranzler HR, O'Malley SS, Gastfriend DR, Pettinati HM, Silverman BL, Loewy JW, Ehrich EW. Vivitrex
Study Group. Efficacy and tolerability of long-acting injectable naltrexone for alcohol dependence: a randomized controlled trial. JAMA
2005;293:1617-1625.
Järvekülg A, Viru A. Opioid receptor blockade eliminates mood effects of aerobic gymnastics. Int J Sports Med. 2002;
23:155-157.
King A, Volpicelli J, Frazer A, and O’Brien C. Effect of naltrexone on subjective alcohol response in subjects at high and low risk
for future alcohol dependence. Psychopharmacology (1997) 129: 15–22.
Kim SW, Grant JE, Adson DE, Shin YC. Double-blind naltrexone
and placebo comparison study in the treatment of pathological gambling. Biol Psychiatry. 2001;49:914-921.
Kim SW. Opioid antagonists in the
treatment of impulse-control disorders. J Clin Psychiatry. 1998;59:159-64.
Murphy MR, Checkley SA, Seckl JR, Lightman SL. Naloxone
inhibits oxytocin release at orgasm in man. J Clin Endocrinol Metab. 1990;71:1056-1058.
Yeomans MR, Gray RW. Opioid peptides and the
control of human ingestive behaviour. Neurosci Biobehav Rev. 2002; 26:713-28.
